A drug developed by researchers in Aberdeen has been put on a fast track programme for approval as trial results show it can slow the progress of Alzheimer’s disease.
TauRX Pharmaceuticals has hailed a “treatment breakthrough” in its third clinicial trial of the drug which involved almost 600 people with the devastating disease.
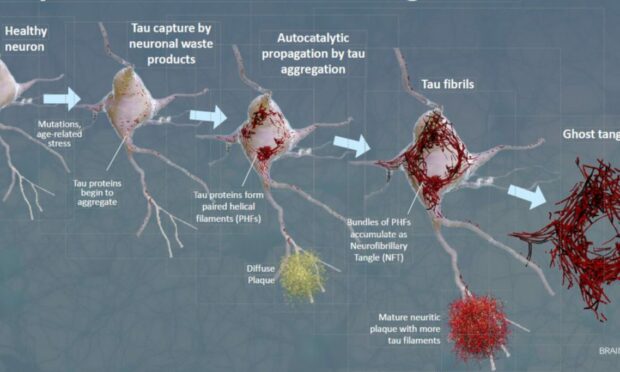
Alzheimer’s disease is the most common cause of dementia and causes people to experience memory loss and other cognitive difficulties.
It is set to affect more than a million Britons by the middle of this century, but its cause and thus its treatment is poorly understood.
The firm’s co-founder Professor Claude Wischik, 72, first observed observed abnormal fibres of protein called tau causing “tangles” in the brains of Alzheimer’s patients over thirty years ago.
Having relocated from Cambridge to Aberdeen University in 1997, Mr Wischik co-founded TauRx along with late Singaporean investor, Dr KM Seng in 2002.
Previous trials of TauRX drugs showed little difference between the drug and a placebo.
In the latest 12-month, phase three trial, participants taking the drug hydromethylthionine mesylate (HMTM) experienced a slowdown in the rate of memory loss and other functional impairmens when compared to other sufferers of the disease.
Our Head of Medical Affairs, Dr Sonya Miller, recently explained what happens in your brain when you have Alzheimer’s Disease, and how current research is making real progress towards a treatment.
Find out more below 🔽#Alzheimers #Dementia #Tau pic.twitter.com/UmMlNmdKNT
— TauRx Therapeutics (@TauRx) May 18, 2022
Professor Wischik, who is a chair in mental health at Aberdeen University, said a full analysis of the data would be released at a later date but he would provide an update on progress at a global conference taking place in London next week.
He said the new drug was a significant innovation for a disease that has not seen any new treatments come to market for decades.
He said: “Today with limited treatments for Alzheimer’s, the standard of care does not impact the underlying causes of symptom progression.
Alzheimer’s remains one of the world’s greatest unmet medical needs.”
Professor Claude Wischik
“HMTM aims to significantly slow disease progression, providing longer term benefits compared to medications brought to market almost twenty years ago.”
He added: “The output indicates that participants receiving HMTM decline at a rate substantially less than is typical in Alzheimer’s based on published research.
“This was seen for both cognitive and functional endpoints across a broad range of severity from mild cognitive impairment (MCI) to moderate Alzheimer’s.
“Importantly, the safety profile is favourable and consistent with previous studies.
“Our data analysis is ongoing and will be reported at a later date.
“We look forward to providing an update on our progress on 9th June 2022 at the 35th Global Conference of Alzheimer’s Disease International.”
On the fast track
Prior to the release of the interim data, the drug was awarded an “innovation passport” by the UK’s drug development regulator in order to speed it through becoming more widely available to people affected by the disease.
Medicines & Healthcare products Regulatory Agency (MHRA) granted the passport which is the first stage of its innovative licensing and access lpathway (Ilap) designed to accelerate development and approval times for new drugs.
Professor Wischik said: “The Ilap designation represents a clear signal of regulatory support for a prospective treatment breakthrough in Alzheimer’s, which remains one of the world’s greatest unmet medical needs.
“Dementia is a leading cause of death around the world, and the innovation passport, as the first stage of the ILAP scheme, enables access to the collaborative approach of regulators and associated health technology assessment bodies to both drug licensing and access throughout the UK.”
Read: Aberdeen researchers developing blood test to show Alzheimer’s risk

Conversation